
OCREVUS is not suitable for everyone, so it is important that you discuss with your doctor or nurse whether this medicine is right for you.
Do not use OCREVUS if: You have had an allergic reaction to OCREVUS or any of its ingredients.
Before you receive OCREVUS, tell your doctor if:
- You have an infection, or a history of recurring or long-term infection (such as hepatitis B).
- You are taking, or have previously taken, medicines which may affect your immune system (such as other medicines for MS).
- You have had, or intend to have, any immunisation with a vaccine.
- You are allergic to any other medicines or any other substances such as foods, preservatives or dye.
- You are pregnant, or intend to become pregnant.
- You are breastfeeding, or plan to breastfeed.
- For more information click here


OCREVUS helps with signs and symptoms of Relapsing-Remitting Multiple Sclerosis (RRMS)
Relapses
When a relapse occurs, symptoms may be experienced suddenly, within a few hours, or slowly develop over a few days. As MS progresses, the damage caused by relapses may grow and could lead to permanent disability.
Remission
In people with RRMS there are periods of full recovery (in which symptoms disappear completely) or partial recovery (in which some symptoms remain).
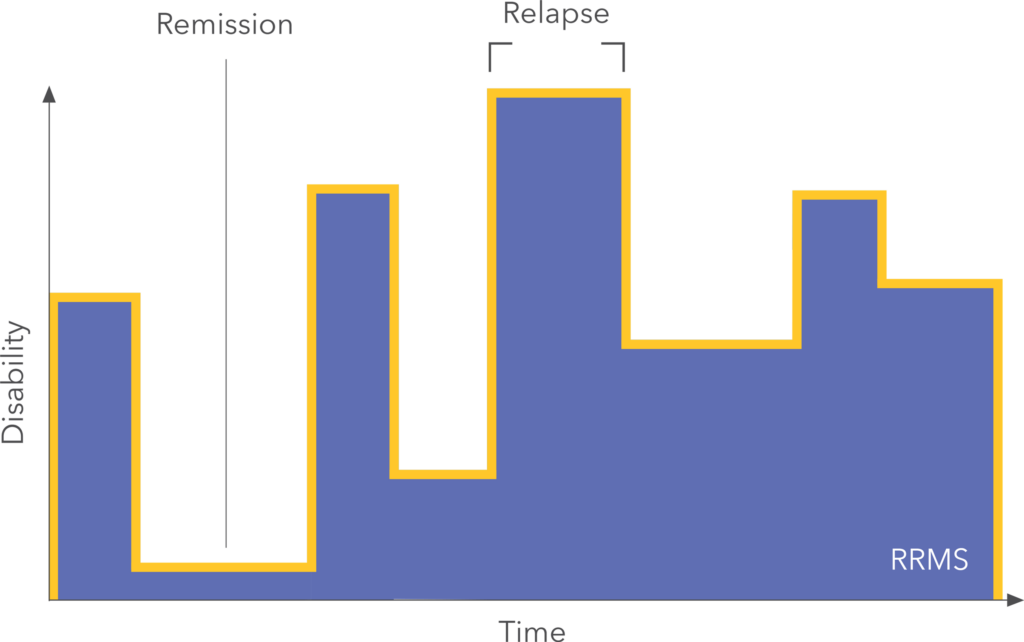
OCREVUS reduces the number or relapses and slows the progression of RRMS compared to beta-interferon (another medication for MS).
OCREVUS has shown promising results so far In reducing the frequency of MS relapses and slowing the overall progression of the disease.


OCREVUS helps with signs and symptoms of Primary Progressive Multiple Sclerosis (PPMS)
PPMS advances more steadily than RRMS.
Symptoms of PPMS continually get worse from the start of the disease. However, while not typical, some people with PPMS may occasionally experience relapses.
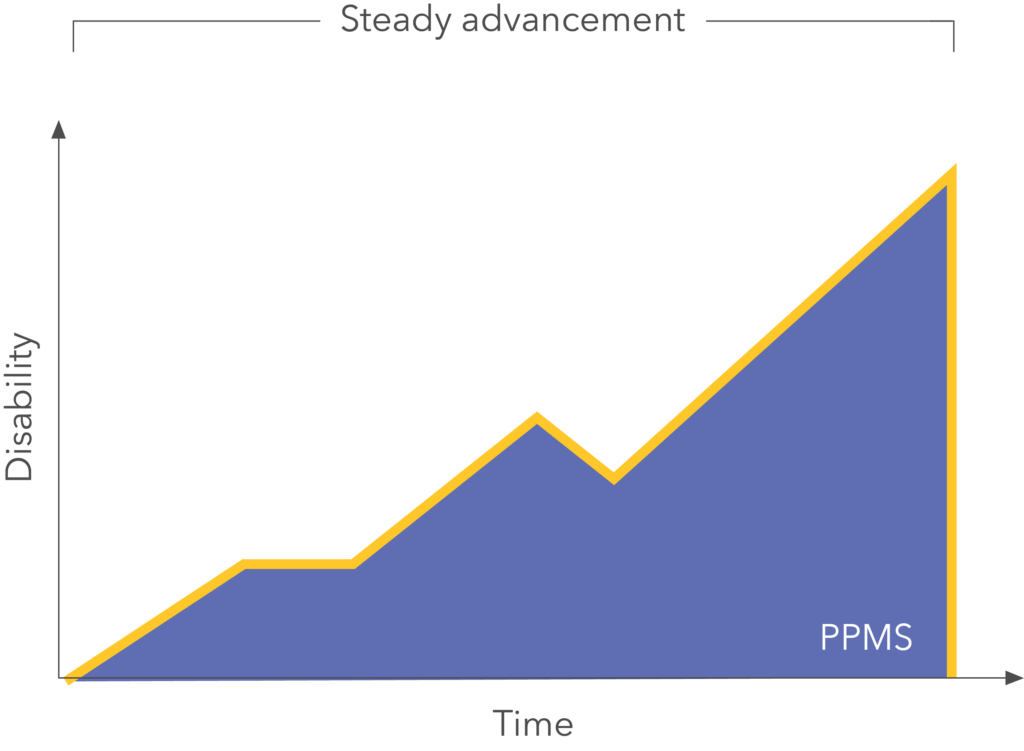
OCREVUS slows the worsening and overall progression of PPMS.


